European Union CE marking Notified Body Construction Products Directive, CARE, miscellaneous, text, trademark png | Klipartz

FIND OUT THE ECM ACCREDITATIONS ON NANDO, THE EU DATABASE OF NOTIFIED BODIES - Ente Certificazione Macchine

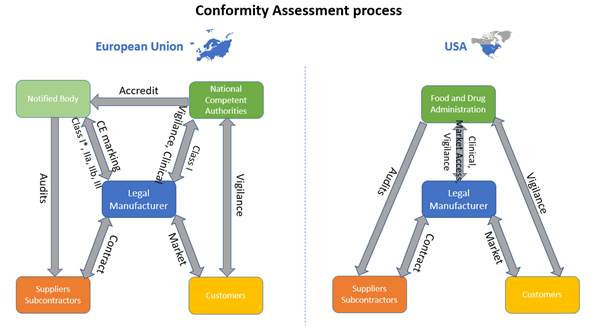
Competent Authority, Notified Body, ISO Registrar: How Each Role Functions in the Medical Device Industry

Mario Gabrielli Cossellu on LinkedIn: #mdr #nando #ivdr #eu #commission #sante #medicaldevices #notifiedbodies… | 11 comments